WHAT IS INFORMED CONSENT?You see an advertisement about a monetary stipend to participate in a revolutionary new genetics clinical trial, and the researchers plan to study your genes. Fantastic! You may get the opportunity to contribute to medicines that change the world for the better. Before you hop into the lab, however, you are presented with a large, dense form detailing the specifics of the study — and a line for your signature at the bottom. What is it, and why does it matter? |
INFORMED CONSENT DEFINED
Informed consent is integral for the ethical conduct of research involving humans, especially in the ever-expanding field of genetics. Based on the ethical principle of respect for persons — the idea that individuals should be treated as autonomous agents — the goal of informed consent is to ensure that subjects are informed enough to make a knowledgeable decision about participating in a research project. |
|
HOW DOES INFORMED CONSENT WORK?
The phrase “informed consent” consists of two equally significant parts. First, the individual must be informed — fully aware of the risks and benefits associated with the research at hand. Second, the individual must provide consent — they must be making a voluntary, willing decision to participate in the research. In order to ensure that informed consent is achieved… |
Research participants must be provided all necessary information so that an individual can make voluntary and knowledgeable decisions about research participation |
2) Informed consent must be accessible and understandable Genomics research requires a clear, specific, and thorough explanation about how the participant’s genomic information will be used, before and after the study |
3) Research participants must be provided all necessary information so that an individual can make voluntary and knowledgeable decisions about research participation Informed consent must be accessible and understandable Genomics research requires a clear, specific, and thorough explanation about how the participant’s genomic information will be used, before and after the study |
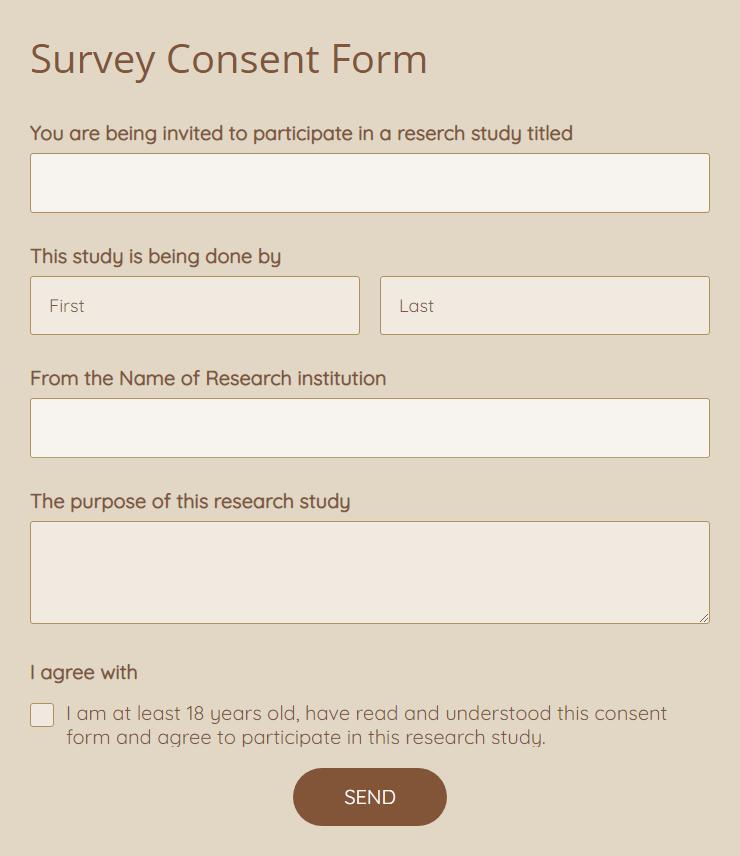
WHAT DOES INFORMED CONSENT LOOK LIKE?
Consent forms are some of the most critical and common components of the informed consent process. They detail the study itself, explain potential effects, and elaborate on an individual's rights as a research participant. The forms are written in simple, non-scientific language, and oftentimes scientists are open to answering questions and hosting information sessions. When filling out a consent form, keep an eye out for... |
|
Risks and discomforts
Benefits Explanation of payment for participation If there will be associated audio/video recordings Use of Tissue Samples/DNA for Future Studies Data sharing Statement of consent |
|
But science, and genetics in particular, can be dense and hard to understand. That is why it is important for researchers to engage in proper science communication practices, while also making sure that all individuals are given adequate time to make their decisions.
EXCEPTIONS
An IRB is a group of five or more people authorized to review, monitor, and approve research involving human participants.
|
|
It is always important to do your own research/brief review about what you are getting involved in.
ADDITIONAL RESOURCES
https://www.genome.gov/about-genomics/educational-resources/fact-sheets/the-informed-consent-process
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3216676/
https://researchservices.cornell.edu/forms/irb-consent-form-templates
https://www.genome.gov/about-genomics/educational-resources/fact-sheets/why-is-informed-consent-required
https://www.michigan.gov/documents/InformedConsent_69182_7.pdf
https://jamanetwork.com/journals/jamapediatrics/fullarticle/485736
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3216676/
https://researchservices.cornell.edu/forms/irb-consent-form-templates
https://www.genome.gov/about-genomics/educational-resources/fact-sheets/why-is-informed-consent-required
https://www.michigan.gov/documents/InformedConsent_69182_7.pdf
https://jamanetwork.com/journals/jamapediatrics/fullarticle/485736
EXTRA READING
Informed consent is the product of many controversial studies and key ethical turning points in history.
During World War II, Nazi doctors engaged in brutal, nonconsensual research on human subjects. This led to the Nuremberg Trials, which led to the Nuremberg Code. They listed 10 ethical principles for scientists to follow, and thankfully, informed consent was one of them.
During World War II, Nazi doctors engaged in brutal, nonconsensual research on human subjects. This led to the Nuremberg Trials, which led to the Nuremberg Code. They listed 10 ethical principles for scientists to follow, and thankfully, informed consent was one of them.